the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
A new bizarre dysoneurid species (Insecta, Trichoptera) in Burmese amber
Wilfried Wichard
Christian Neumann
We describe an extinct caddisfly species, Cretapsyche palpinova sp. nov., from mid-Cretaceous Burmese amber. The species belongs to the extinct family Dysoneuridae within the superfamily Sericostomatoidea. The family includes some species with unusual maxillary palps. Cretapsyche palpinova sp. nov. is characterized by five-segmented maxillary palps, the second segment being the longest, and the third segment bearing an unusual pin-shaped appendage.
The extinct trichopteran (caddisfly) family Dysoneuridae was established by Sukatsheva (1968). The description is merely based on fossil wings, which are preserved as impressions in sedimentary rocks from the Upper Jurassic of the Karatau Mountains in southern Kazakhstan. Sukatsheva and Vassilenko (2013) once again described the family using further fossil wing impressions found at various Eurasian sites from the Middle Jurassic to the Lower Cretaceous.
The preliminary diagnostic forewing characters consist of the absence of apical forks I and IV; the radius R thickened, running straight, without bifurcation; the discoidal and medium cells closed or open; the thyridial cell closed and long; Cu2 reaching a desclerotized crossvein running between Cu1b and the wing margin.
The description of the family Dysoneuridae was reviewed when new and complete specimens were found in mid-Cretaceous Burmese amber. An expanded set of important morphological characters were added to the preliminary description of the family. Fore- and hind wings are often excellently preserved, as well as the head, thorax, and abdomen including antennae, the maxillary and labial palps, the compound eyes and the ocelli, the legs with characteristic spurs, and the genitalia (Wichard et al., 2018).
The amber bearing the embedded caddisfly belongs to Burmese amber from northern Myanmar. Most Burmese amber comes from the amber mine located near Noije Bum Village, Tanaing Town (Kania et al., 2015: fig. 1; Jarzembowski et al., 2017: Fig. S1). The age provided by U–Pb dating of zircons from the volcaniclastic matrix of the amber is early Cenomanian (98.8±0.6 Ma) (Shi et al., 2012).
The fossil specimen represents a nearly complete adult, visible in ventral and dorsal views. The hind wings are partially covered by the forewings. The male genitalia are visible in ventral view.
Photos were taken using a Leica stereomicroscope M 420 Apozoom in combination with a Canon EOS 80D DSLR, EOS utility software, and Zerene Stacker software. All images and figures were processed with Adobe Photoshop CS4 and Photoshop Elements 15.
Abbreviations
Wing venation terminology generally follows Holzenthal et al. (2007):
I denotes apical fork I, II denotes apical fork II, III denotes apical fork III, V denotes apical fork V, R denotes radius, Rs denotes radius sectori, M denotes media, Cu1 denotes cubitus anterior, Cu2 denotes cubitus posterior, A denotes analis, DC denotes discoidal cell, MC denotes median cell, and TC denotes thyridial cell.
In male genitalia, inf ap denotes inferior appendages, pre ap denotes preanal appendages, and X ap denotes mesodorsal processes of tergum X.
This published work and the nomenclatural acts it contains have been registered in ZooBank on 26 July 2019 and have received the LSID number urn:lsid:zoobank.org:act:1161465B-1937-4580-B02F-8ED51C484A79.
Systematic paleontology
-
Order Trichoptera Kirby, 1815
-
Suborder Integripalpia Martynov, 1924
-
Family Dysoneuridae Sukatsheva, 1968
-
Type genus and species: Dysoneura trifurcata Sukatsheva, 1968.
Family diagnosis
Dysoneuridae clearly distinguished from all extant and extinct families of Trichoptera by the tibial spur formula 2/2/4 in combination with the following fore- and hind wing venational characters. In forewing, forks I and IV absent and forks II, III, and V present; the nygma absent; discoidal and median cells closed or open; the thyridial cell long and closed; Cu2 reaches the desclerotized line (crossvein) running from the wing margin to Cu1b. In the hind wing, forks I, III, and V are present; and forks II and IV and the discoidal cell are absent. Antennae about as long as the forewings or longer; the scapus as long as the head or longer, often bearing a brush of long dark setae.
The family Dysoneuridae belongs to superfamily Sericostomatoidea indicated by the absence of ocelli, with the terminal maxillary and labial segments being nonannulated and inflexible and the tibial spur formula being 2/2/4. The Dysoneuridae are distinct from all other families of Sericostomatoidea by their typical wing venation of fore- and hind wings.
Remarks
The family Dysoneuridae as established by Sukatsheva (1968) is based merely on the venation of an isolated forewing preserved in sedimentary rocks from the Upper Jurassic in southern Kazakhstan. The forewing venation was interpreted as a reduced venation because of the absence of apical forks I and V; in addition, Cu2 reaches a desclerotized line running from the wing margin (arculus) to Cu1b; both characteristics correspond approximately to those of the wings of the family Kokiriidae (Sukatsheva and Vassilenko, 2013). However, for a complete and significant description and classification of Trichoptera, a complete set of morphological characters is imperatively needed. In the case of the family Dysoneuridae, this became recently available by excellently preserved specimens from Cretaceous Burmese amber, subsequently leading to a redescription of the family Dysoneuridae and their assignment to the superfamily Sericostomatoidea (Wichard et al., 2018).
-
Cretapsyche Wichard et al., 2018
-
Type species: Cretapsyche circula Wichard et al., 2018
-
Genus diagnosis: the five-segmented maxillary palps give a clear and striking characteristic – second segment longest, longer than the following three segments together, which are successively smaller and shorter; fifth segment shortest.
-
Cretapsyche palpinova sp. nov.
-
Figs. 1–2.
Holotype
Kept in the amber collection of the Museum für Naturkunde, Berlin (MfN: holotype inventory number MB.I. 7340 coll., former Patrick Müller collection, BUB 2587). Fossil from Upper Cretaceous, Cenomanian. Embedded in Burmese amber, body well preserved; forewing in dorsal view, hind wing covered dorsally by forewing. Antennae and legs present. Male genitalia visible in ventral view.
Etymology
Cretapsyche palpinova sp. nov. is named for its novel maxillary and labial palpi.
Diagnosis
Scapus about as long as the head and bearing a brush of long setae; the pedicellus short, thickened at end; first flagellomere almost twice as long as the following flagellomeres and basally thickened. The new species is distinguished from other dysoneurid species by the maxillary and labial palps: maxillary palps five-segmented; second segment longest, narrow, and slightly bent; third segment hairy, pin-shaped pendant (pilifer); third to fifth segments successively decreasing in size. Labial palps three-segmented; the terminal segment long, oval, and flattened, with an apicolateral bulge.
Description
Male head without ocelli. Each flagellum about 4 mm in length, consisting of 26 rod-shaped flagellomeres; scapus strong, narrow, distally slightly broader than basally, about 0.5 mm in length and slightly longer than head, hairy, apically with tuft of hair; pedicellus short, thickened at ends; first flagellomere twice as long as following flagellomeres, basally thickened. Maxillary palpus five-segmented: first segment long but shorter than second segment; second segment longest, slim and slightly curved, longer than third, fourth, and fifth segments together. Rod-shaped appendage (pilifer) originating from third segment. Labial palpus three-segmented: first segment longest, almost as long as second and third segments together; third segment about as long as second segment but wider, flattened, oval, rounded margin, apicolaterally with small rounded tongue.
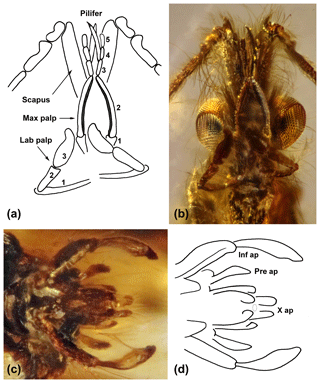
Figure 2Cretapsyche palpinova sp. nov. (a) Drawing of the scapi, pedicelli, and first antennal segments; the five-segmented maxillary palps (max palp), the three-segmented labial palps (lab palp); and a pair of pilifers arising from the maxillary palps. (b) Head with palps and antennae in ventral view. (c) Male genitalia in ventral view. (d) Drawing of the male genitalia in ventral view with the inferior appendages (inf ap) consisting of the basal coxopodite and the apical harpago, with the preanal appendages (pre ap) and with tergum X divided in two apical lobes (X ap).
Forewings each 3 mm in length (Fig. 1b). Apical forks I and IV absent; II, III, and V present; fork III petiolate; fork V probably slightly petiolate. Crossveins r and s not clearly visible, r-m short and aligned diagonally, m absent. Discoidal and median cells open, thyridial cell long and closed. Cu2 running to Cu1b and reaching desclerotized crossvein connecting Cu1b to margin; this crossvein not clearly visible. Hind wings smaller than forewings, each with wing venation and setation as follows (in ventral view, so far as visible): fork III clearly visible, petiolate; crossvein r-m also clearly visible; fork II absent; forks I and V probably present; dorsal surface covered with small scale-shaped hairs.
Male genitalia visible in ventral view (Fig. 2c, d). Inferior appendages two-segmented, each consisting of coxopodite and harpago; small subtriangular accessory appendage basoventrally; coxopodite strong, about 0.2 mm in length, about as long as harpago; harpago originating from coxopodite, basally narrow, thicker mesally near middle, and tapered apically; curved mesad, enclosing central phallic apparatus. Tergum X with mesodorsal lobe divided apparently into two small parallel processes covering phallic apparatus dorsally. In ventral view dorsolateral preanal appendages also deeply behind central phallic apparatus, each forming small, elongate, triangular spatula.
In general fossil insects in Burmese Amber exhibit a surprisingly great range of variations and morphological characteristics, which are often unknown to today's insects. This also applies to the Trichoptera. A comparison of the recent caddisflies with the fossil caddisflies embedded in the 40- to 50-million-year-old Baltic Amber points to clear differences but also shows an amazing closeness and relationship to today's forms (Ulmer 1912, Wichard 2013). Even clearer are the differences compared to the almost 100-million-year-old caddisflies preserved in Burmese amber. Of course, with their greater age, Burmese Trichoptera are more distantly related to recent caddisflies. However, morphological variants were also tested, which obviously did not survive in the course of time and evolution, so that modern species lack them.
As proposed by Wichard et al. (2018), the extinct family Dysoneuridae belongs to the superfamily Sericostomatoidea, characterized by the absence of ocelli, with the terminal maxillary and labial segments both being nonannulated and inflexible, and with the tibial spur formula being 2/2/4 (Johanson et al., 2017). The family Dysoneuridae is distinguished from all other families of Sericostomatoidea by its wing venation with the absence of forks I and IV in the forewings and of forks II and IV in the hind wings.
The Dysoneuridae found in mid-Cretaceous Burmese amber consist of two interesting genera, Burmapsyche and Cretapsyche. Burmapsyche has maxillary palps which are three-segmented. In B. palpifurcata each palpus is forked into a one-segmented branch and a two-segmented branch, an unusual small fork, unknown in modern caddisflies. The genus Cretapsyche is characterized by five-segmented maxillary palps, equipped with a remarkably long, slightly curved second segment. Additionally, in Cretapsyche palpinova a pin-shaped appendage originates from the third segment of each maxillary palpus that slightly towers above the palpus distally.
(Family and genus characters: five-segmented maxillary palps each with second segment longest; in forewing forks II, III, and V and in hind wing forks I, III, and V present; tibial spurs 2/2/4).
-
In forewing, Rs and M branching on about the same level, fork V petiolate; in male genitalia coxopodite and harpago at a ratio of about 1:1 in length: 2
- –
In forewing, M branching before Rs, fork V nearly sessile; in male genitalia coxopodite and harpago at a ratio of about 2:1 in length: 3
- –
-
Labial palps three-segmented, terminal segment oval flattened; maxillary palps with pilifer arising from third segment: C. palpinova sp. nov.
- –
Labial palps three-segmented, simple; maxillary palps without pilifer on third segment:
C. circula.
- –
-
Forewing with median cell open; in male genitalia dorsomedian process of tergum X distinct, ventrally protruding: C. insueta.
- –
Forewing with median cell closed; in male genitalia dorsomedian process of tergum X simple:
C. elegans.
- –
The material described herein is deposited in the amber collection of the Museum für Naturkunde, Berlin (inventory number MB.I.7340).
WW and CN prepared the paper and contributed to the editing.
The authors declare that they have no conflict of interest.
We especially thank Patrick Müller for the excellent collection of Burmese amber inclusions and for making this newly described species available. Further, we wish to express our sincere gratitude to John Morse (Clemson University, USA) and Bo Wang (Academy of Sciences, Nanjing, China), who provided critical reviews that helped in improving the present contribution of the manuscript.
This paper was edited by Florian Witzmann and reviewed by Bo Wang and John C. Morse.
Holzenthal, R. W., Blahnik, R. J., Prather, A. L., and Kjer, K. M.: 2007. Order Trichoptera Kirby, 1813 (Insecta), Caddisflies, Zootaxa, 1668, 639–698, 2004.
Jarzembowski, E. A., Wang, B., and Zheng, D.: A new ommatin beetle (Insecta: Coleoptera) with unusual genitalia from mid-Cretaceous Burmese amber, Cretaceous Res., 71, 113–117, 2017
Johanson, K. A., Malm, T., and Espeland, M.: Molecular phylogeny of Sericostomatoidea (Trichoptera) with the establishment of three new families, Syst. Entomol., 42, 240–266, 2017.
Kania, I., Wang, B., and Szwedo, J.: Dicranoptycha Osten Sacken, 1860 (Diptera, Limoniidae) from the earliest Upper Cretaceous Burmese amber, Cretaceous Res., 52, 522–530, 2015.
Kirby, W.: Strepsiptera, a new order of insects proposed, and the characters of the order, with those of its genera, Transactions of the Linnean Society of London Zoology, 11, 86–122, 1813.
Martynov, A. V.: Rucheiniki (caddisflies), Prakticheskaya Entomologiya, 5, 1–384, 1924 (in Russian).
Shi, G., Grimaldi, D. A., Harlow, G. E., Wang, J., Wang, J., Yang, M., Lei, W., Li, Q., and Li, X.: Age constraint on Burmese amber based on U-Pb dating of zircons, Cretaceous Res., 37, 155–163, 2012.
Sukatsheva, I. D.: New Jurassic Caddisflies from the Kara Tau (Trichoptera), Yurskie nasekomye Karatau, 175–179, 1968 (in Russian).
Sukatsheva, I. D. and Vassilenko, D. V.: New Taxa of Caddisflies (Insecta, Trichoptera) with reduced forewing venation from the Mesozoic of Asia, Paleontologicheskii Zh., 2013, 57–63, 2013.
Ulmer, G.: Trichopteren des Baltischen Bernsteins, Beiträge zur Naturkunde Preussens, 10, 1–380, 1912.
Wichard, W.: Overview and descriptions of Trichoptera in Baltic Amber: Spicipalpia and Integripalpia, Kessel, Remagen, Germany, 230 pp., 2013.
Wichard, W., Neumann, C., Müller, P., and Wang, B.: Family Dysoneuridae (Insecta, Trichoptera) in Cretaceous Burmese amber, Cretaceous Res., 82, 138–146, 2018.





